Spastic paresis is a frequent complication of lesions to central motor pathways including the upper motor neuron, and is deeply distressing for patients, not only because of the physical effects on their bodies but also due to the emotional impact of the lack of control of limb movements that may deprive them of their independence.1 Patients who develop spasticity require ongoing rehabilitation programmes, initiated as early as possible, to alleviate their symptoms, help them to relearn motor skills and, ultimately, to regain at least some of their independence.2 Patients’ expectations of treatment often focus on their need for greater function (and hence independence).3 They may wish to be able to let themselves into the house unaided, to be able to drive or to reduce the frequency with which they have to replace their shoes due to wear.
Typically, rehabilitation programmes are now based on intramuscular injections of botulinum toxin and muscle stretching and training exercises to overcome paresis. Recent clinical studies have suggested that intensive exercise in long-term guided self-rehabilitation programmes (in which the patient is coached by an expert therapist but works mainly on his or her own) may significantly improve outcomes for patients.3,4
Patient-orientated goal setting and attainment scaling (GAS) can be used to align physicians’ and patients’ objectives for rehabilitation to tailor these to the individual patient’s needs.5,6
The Comprehensive Care of Patients with Spastic Paresis: A Long-Term Commitment symposium, presented as part of the International Society of Physical and Rehabilitation Medicine World Congress in Berlin in July 2015, explored best practice in the long-term management of patients with spastic paresis. There are three pillars that support the management process in these patients:
• patient-centred goal setting
• detailed clinical assessment covering all aspects of the rehabilitation process
• an active role for patients in treatment, notably through guided selfrehabilitation programmes.
Success in all three of these areas is likely to keep the patient engaged and motivated, and ultimately to lead to patient and clinician satisfaction with treatment.
Agreeing Treatment Goals with Patients – The First Step towards Patients’ and Clinicians’ Satisfaction
The inclusion of the patient’s perception of success with regard to treatment goals represents a paradigm shift in medicine. It acknowledges that – especially in terms of chronic diseases – the impact of disease can go much further than measurable clinical changes in the body, and can affect patients’ self-esteem, mood, interactions with family and society in general and overall quality of life.7,8
In order to improve the chance that treatment outcomes will meet patient expectations, it is important for both clinicians and patients to agree on the goals of treatment and the timescale for achieving them. GAS is a helpful tool for this individualised approach because each scale is specific to the individual and the defined endpoints are related to health goals.5 GAS uses a five-point scale to assess achievement of goals (from -2 to +2). This method, first introduced by Kiresuk and Sherman in 1968,9 is increasingly used (alongside established outcomes measures) in the management of spastic paresis.10–12
The first step in the GAS process is to establish which goals are important to the patient and which symptoms have the greatest possibility of improvement. This process can be challenging even for experienced therapists and requires the patient to be fully engaged and without cognitive deficit or depression for success. This requires honest and frank discussions between the clinician and patient, and possibly other members of the rehabilitation team, the patient’s family, employers and organisations funding the rehabilitation process.10 Clinicians should bear in mind that a recent global survey of patients living with spasticity revealed 44% of respondents had not discussed their expectations of treatment with their doctor.13 The clinician therefore needs to facilitate this discussion.
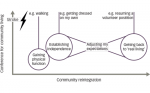
The results of these discussions should be SMART goals14 – i.e. goals that are specific (clear and precise), measurable (detailed enough to allow objective assessment of goal attainment), achievable (failure to achieve a goal can have a negative effect on motivation), realistic (taking into account the time, resources and support required) and timed (set a target date – e.g. within 3 months – and revise and update the goal when the date is reached)15 (see Figure 1).
Patients with spastic paresis are likely to set a combination of active and passive goals to achieve increased function. Active goals may include improvements in activities of daily life (washing, dressing, cooking, working), or walking faster and/or further. Passive goals may include being able to dress and maintain good hygiene for the affected limb. Additional goals may include reductions in contractures, spasms and pain.16
In order to be SMART, goals need to be positive, such as being able to walk a particular distance, or perform a particular activity.

Once the goals have been agreed, the next step is to plan how attainment is to be achieved and measured. A number of scales exist that can be used to measure changes in active and passive function (Arm Activity [ArmA] scale, Numeric Graphic Rating Scale [NGRS], 10-minute walk test), severity of impairment (Neurological Impairment Scale [NIS], Apparent Affect Rating Scale [AARS]), and pain and discomfort (Scale of Pain INtensity [SPIN], NGRS) in addition to simple measures of joint angle, distance from fingertip to palm, time taken to achieve a task and numbers of nocturnal awakenings.11,17
After treatment, patients and clinicians should monitor progress towards each goal at the target date. GAS should be an iterative process – if a goal is not attained, the method and/or the target date for reaching the goal should be revisited and a new target date agreed.
Goal Setting and Attainment Scaling in Practice – Evidence from the Upper Limb International Spasticity Programme
The Upper Limb International Spasticity (ULIS) programme (see Figure 2) was initiated in 2007,18 and is the first large international real-life study to focus on the evaluation of patient goals. The programme consists of a series of observational studies designed to describe current clinical practice in the application of BoNT-A in the management of upper limb spasticity.
The first stage of the programme documented clinical practice by evaluating treatment and outcome evaluation around the world.18,14
The second stage, ULIS II, was a large, before-and-after, prospective, observational cohort study that included GAS as primary outcome measure following one cycle of BoNT-A and it was used to assess overall

attainment of treatment goals.14 Achievement of primary and up to three secondary SMART goals – as measured by appropriate assessment scales (selected by the clinician) – were rated using the simplified ‘GAS-light’ approach developed by Turner-Stokes. With GAS-light, clinicians use words instead of numbers to describe the patient’s progress and then use an algorithm to create a score.5 The data collected in ULIS II confirmed the efficacy of BoNT-A in patients with upper limb symptoms and on person-centred outcomes relating to passive/active functions and to impairment/symptoms.
In the next step of the programme, ULIS III aims to expand the cohort and record information over several cycles of BoNT-A treatment and assess the benefits of integrated spasticity management.
Summary
Goal setting is important to engage and motivate patients to persist with their treatment programme. The process should begin with defining and agreeing SMART goals that are meaningful for and achievable by the patient and should be iterative – if the patient does not achieve a goal at the first attempt, the programme should be revised to help the patient achieve the goal at a subsequent attempt.
How Can Clinical Assessment Scales Influence Patient Management?
There are two basic types of scale used for assessing and monitoring change in spasticity: impairment scales (such as the Modified Ashworth Scale [MAS], the Tardieu scale) and functional scales (such as the Modified Frenchay Scale [MFS], Disability Assessment Scale [DAS], Barthel Ind ex) and GAS.5,19–21
The MAS has been used to assess the severity of spasticity in many clinical studies as well as in everyday practice.19 The MAS actually measures a combination of soft tissue contracture, spastic dystonia and spasticity, and so is not always the most accurate scale for assessing spasticity. Moreover, between segment comparisons are difficult because each has a different range of motion; this means that the velocity of passive muscle lengthening (and therefore impact of spasticity) is variable.21–23
While not as widely used as MAS, the Tardieu scale, which was created in 2000 after Tardieu’s clinical method of child examinations in the 1960s, is more specific for assessing spasticity.24 It measures resistance to movement using the angle of arrest: in slow passive motion (V1) to assess spastic dystonia and contracture; and at very fast speed (V3) to assess spasticity and contracture. The difference between V3 and V1 has been termed the spasticity angle, measuring spasticity in a way that is minimally dependent on muscle length.14,22 The grade of spasticity is scored on a scale of 0 to 4, ranging from no resistance to unfatiguable clonus.21
Functional scales are used in association with impairment scales to provide a broader assessment of the patient’s specific needs in daily life. It should be noted that functional clinical scales are also important psychotherapeutic tools, which help maintain the motivation for therapy not only in the patient and his/her family, but, in fact, in the whole therapeutic team.
Clinical Assessment Scales in Practice – The AbobotulinumtoxinA Adult Upper Limb Phase III Study
Although it is known that botulinum toxin can produce positive outcomes on function, pain and overall goal attainment by decreasing muscle tone and spasticity,25 no studies have demonstrated effects on all of muscle tone, spasticity, active movement and function in hemiparetic patients. The AbobotulinumtoxinA Adult Upper Limb (AUL) study was a three-arm, international, phase III, randomised, placebocontrolled, double-blind trial in 34 centres across nine countries. It was designed to assess the efficacy and safety of abobotulinumtoxinA (Dysport; Ipsen) in 243 adults with hemiparetic upper limb spasticity due to stroke or traumatic brain injury. Patients were randomised to 500 or 1,000 mU abobotulinumtoxinA or placebo injected into the primary targeted muscle group (the group with the highest MAS at baseline, as determined by the investigator) and were followed for up to 24 weeks.26,27
This study used a combination of the MAS, Tardieu and DAS, in addition to Physician’s Global Assessment (PGA), active range of motion measurements (AROM) and ease of applying a splint to assess modifications in muscle tone and passive and active movement following treatment with abobotulinumtoxinA.
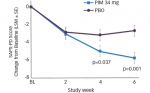
The AUL study demonstrated a significant improvement (p<0.001) in muscle tone (as assessed by MAS) starting in the first week after treatment and persisting for up to 20 weeks in some patients with both doses of abobotulinumtoxinA. There were also clinically meaningful improvements in active range of motion and an objective reduction in spasticity (according to the Tardieu scale) following treatment, in addition to improvements in DAS.26
Summary
The AUL study not only demonstrated the efficacy of both doses of abobotulinumtoxin A in reducing muscle tone in patients with upper limb hemiparesis and adding other clinical benefits, it also showed how the use of different scales for assessing patients, including active range of motion, ease of applying splint, DAS and other scales can optimise the way the toxin is used and provide meaningful results for the patient and information for the treating physician.
Guided Self-Rehabilitation Contracts – A Companion to Botulinum Toxin Therapy in Helping Patients Achieve Treatment Goals
Statistically, patients with spastic paresis undergoing physical therapy often reach a plateau in their progress after about 6–9 months.28,29 At this point, rehabilitation prescriptions are discontinued (or possibly converted into a form of maintenance therapy) for most patients. Only by understanding the processes underlying the development of spastic paresis can we hope to begin to improve this situation.
Spastic paresis superimposes two disorders around each joint: a neural disorder comprising stretch-sensitive paresis in agonists and muscle overactivity in antagonists, and a muscle disorder (spastic myopathy) combining shortening and loss of extensibility in antagonists.30–32
Without therapy, patients enter a downward spiral in which affected limbs are left relatively immobilised, leading to chronic disuse, which adds further paresis as the brain’s ability to voluntarily recruit motor units declines. Clinical assessment of the patient may be a fivestep process beginning with an assessment of limb function (using ambulation speed or MFS scores) and continuing with an evaluation of functional muscle length XV1 measured at slow speed, angle of catch XV3 upon fast stretch, maximal active range of motion XA against the antagonist (capacity to overcome stiffness and spastic co-contraction) and the residual active amplitude after 15 seconds of maximal amplitude rapid alternating movements, XA15. From these measurements, coefficients of shortening and weakness can be calculated, which help tease out the muscle and the neural disorders.32
This distinction between the muscular and neurological disorders is important because the conventional methods of rehabilitation for chronic, severe motor impairment have often insufficiently prevented or treated the muscle disorder in particular. A high coefficient of shortening indicates that the muscular disorder is the priority for treatment, whereas a high coefficient of weakness indicates that the neurological disorder is the priority.21,30–32
Treatment programmes involving rapid alternating maximal efforts or unassisted rapid alternating movements of maximal amplitude, have been shown to attenuate the neurological disorder of motor command in gradually reducing co-contraction by restoring reciprocal inhibition between antagonists during motor command.33,34
It has also been shown that prolonged daily self-administered stretch postures may reduce symptoms of spastic myopathy by gradually lengthening muscles and reducing their stiffness and spindle sensitivity.35 Evidence shows that patients who are given exercises to continue at home value them for the structure they can give to the day and for offering hope that, with hard work, ongoing reductions in spasticity can be achieved.3,4 It is important for the clinician to work with the patient to manage these expectations, however.
Recently, a system of guided self-rehabilitation contracts (GSCs), consisting of prolonged daily self-administered stretch postures to gradually lengthen muscles and reduce their stiffness and spindle sensitivity, has been developed36 as a strategy to produce or increase patient and therapist motivation to avoid plateauing. GSC render the combination of daily intensity and long-term duration of the rehabilitation work realistic for patients, clinicians and payers.
Guided Self-Rehabilitation in Practice
An ongoing multicentre study is comparing outcomes from the GSC programme with those from conventional community-based physical therapy. For those in the GSC arm, a contract setting out what the patient needs to do is required for each affected joint. The timescale for the patient to achieve the contract needs to be realistic. Patients following GSCs were provided with a multi-lingual application for tablets and cell-phones in several countries to increase longterm patient engagement and to facilitate monitoring, provision of programme adjustments and other communications between the patient and the therapist.37
Summary
The onset of muscle shortening can occur within hours of brain injury. Timely initiation of therapy can help to prevent muscle symptoms. Exercises that target both the muscular and neurological components of paresis can improve function and quality of life. GSCs may include challenging muscle stretches that are repeated many times for at least 30 minutes a day, alternating with bouts of rapid alternating movements of maximal amplitude against the stretched muscle. Only patients have the time to devote to performing these exercises and they should be encouraged to continue exercising by a qualified therapist acting as a coach, by the use of daily diaries that help the patients to see what progress they are making and potentially using a smartphone app to provide reminders and encouragement to patients to persist with the programme.
Conclusion
There is growing acceptance that the needs of patients with spasticity are best addressed by taking a comprehensive approach to their management – i.e. one that encompasses the patient’s physical needs and mental wellbeing, beginning at the first assessment and continuing through the various stages of the rehabilitation process. The use of GAS can engage and motivate patients to play a full part alongside their care teams. Appropriate clinical assessment scales provide meaningful information for the physician in regards to the complete treatment plan, including BoNT-A and a rehabilitation program. Guided selfrehabilitation might help to achieve the best possible outcomes.