Defining Pain
Defining Pain
Pain has been defined as an ‘unpleasant sensory experience associated with actual or potential tissue damage or described in terms of such damage’. This is the official definition of the Taxonomy Committee of the International Association for the Study of Pain.1 Under normal circumstances, pain is a normal physiological response experienced when peripheral sensory units (called nociceptors) are activated and transduct noxious, or potentially noxious, energies into action potentials that are transmitted to the central nervous system.2 This is what is called nociceptive pain, and can be successfully treated by analgesic agents such as non-steroidal anti-inflammatory drugs or opioids.
Pain may also occur when there is a ‘primary lesion or dysfunction’ in the peripheral or central nervous system. This type of pain is called neuropathic pain, and is commonly unresponsive to classical pain drugs.1 Neuropathic pain may have multiple origins, for example mechanical nerve damage (e.g. amputation, tumours), metabolic disturbances (e.g. diabetes), infection (e.g. postherpetic neuralgia or human immunodeficiency virus (HIV) neuropathy) or it may be of central origin (e.g. stroke, spinal cord injury or multiple sclerosis). A number of patients complaining of pain have co-existence of inflammatory/nociceptive pain and neuropathic pain. These pain states have been called ‘mixed pain’. The boundaries of neuropathic pain are thus sometimes blurred. Therefore, it is important to diagnose and recognise neuropathic and mixed pain in order to ensure that patients receive the most appropriate treatments.
Dysfunction of the nervous system may produce negative symptoms (for example a paralysis) or positive symptoms (for example pins and needles, tingling or burning pain). Patients with neuropathic pain will always present with a variety of negative and positive symptoms.3 Neuropathic pain is a complex group of positive sensory phenomena that are experienced as painful by patients. Complications arise because each particular positive sensory symptom has a discrete pathophysiology, which may respond differentially to treatments targeting it. Therefore, it is of crucial importance to obtain agood understanding of the different pathophysiological mechanisms behind each of the symptoms and signs that constitute neuropathic pain.
Stimulus-independent and Stimulus-evoked Pain
Neuropathic pain can manifest itself as either spontaneous pain, elicited without need of a stimulus (stimulus-independent pain) and/or as pain hypersensitivity elicited after a stimulus (stimulus-evoked or stimulus-dependent pain).
Stimulus-independent pain includes symptoms that are experienced by patients spontaneously, and may involve different types of abnormal sensations such as continuous burning sensation, intermittent shooting, lancinating sensations, electric-shock-like pain, some paresthesias, or dysesthesias (i.e. pins and needles, tingling). Stimulus-evoked pain comprises hyperalgesia and allodynia, which may be elicited after mechanical, thermal or chemical stimulation.1 In most patients with neuropathic pain there is a combination of stimulus-evoked signs and stimulus-independent symptoms (see Table 1).
Mechanisms of Neuropathic Pain
The basic function of axons is nerve conduction.4 For the system to be reliable, conduction must be accomplished both without loss and without gain. Impulse conduction without loss means that if a single action potential is generated at the receptor level, a single action potential must reach the spinal cord. If the impulse is lost during transmission, then a sensory deficit will occur. This is the cause of negative phenomena. Accordingly, impulse conduction without gain means that if a single action potential is generated at the receptor level, not more than one action potential reaches the spinal cord. Generation of extra impulses during conduction would distort the sensory message. In general, the precipitating cause of neuropathic pain is the generation of abnormal impulses in the somatosensory system.5-9 Axons that were initially only conductors of impulses become, due to hyperexcitability, generators. In other words, increased membrane excitability may lead to ectopic impulse generation in damaged axons. Ectopic impulses can be, and in the majority of cases will be, transmitted along normal central pathways to reach brain areas and eventually elicit a conscious perception.10
Ectopically generated impulses are known to set up a state of increased sensitivity in central nervous system sensory neurons.11 This has been called ‘central sensitisation’. During recent decades, much of the research and industry efforts have been invested in trying to understand, and eventually target, the mechanisms behind central sensitisation. A key target has been the N-metyl-D-aspartate (NMDA) receptor and its role in setting up central sensitisation.12 It has been hypothesised that modulating this receptor could revert, or at least control, central sensitisation, and therefore reduce pain. However, despite countless efforts, the practical results of this line of research have been rather poor.13 Currently, there is no single NMDA antagonist widely used to treat neuropathic pain.
Importance of Ectopic Impulse Generation
As mentioned earlier, neuropathic pain is not a single concept, but a complex group of positive sensory phenomena due to abnormal function at different levels of the nervous system. Each of these abnormal sensory phenomena may be due to a discrete pathophysiological mechanism operating at a defined level. There exists well-established evidence for some of these mechanisms. For example, there is good evidence that the pathophysiological mechanism of a Tinel’s sign, the bothersome increased mechanosensitivity of damaged peripheral nerves, is due to an abnormal accumulation of mechanosensitive sodium channels (of yet unknown type). However, for the vast majority of other symptoms/signs that form the ‘neuropathic pain complex’ there are scarce, and sometimes conflicting, proposed mechanisms. This is particularly true for the so-called evoked phenomena, namely hyperalgesia and allodynia.
Although current emphasis is put on possible mechanisms of central hyperexcitability and sensitisation, there is increasing awareness that peripheral mechanisms operating at the level of the damaged axonal membrane are at the core of neuropathic pain symptoms.14 There are straightforward observations to support this claim. For example, it is a common experience that all positive sensory phenomena, spontaneous or evoked, due to a focal peripheral nerve injury, immediately disappear when a few millilitres of lidocaine are injected in its close vicinity. This seems to indicate that the initiating and relevant processes, ultimately leading to the experience of neuropathic pain, are generated at this peripheral level. There is ample consensus that states of central hypersensitivity are generally the consequence of an altered peripheral input, i.e. ectopic impulses from damaged sensory axons. Therefore, it is reasonable to explore the mechanisms of the hyperexcitability that lead to ectopic impulse generation in peripheral sensory neurons, and how they link to the human experience of neuropathic pain.
Mechanisms of Axonal Hyperexcitability
One of the key steps in the study of the mechanisms of hyperexcitability is to understand how a region of the axonal membrane changes its properties from a mere conductor to a generator of impulses, both spontaneously or in response to mechanical, thermal or biochemical changes in the microenvironment surrounding it.15
It is beyond the scope of this review to address all the molecular changes occurring in damaged neurons that will ultimately render them hyperexcitable. It is known that the physiological pattern of ion channel expression changes in neuropathic pain conditions.16 These changes affect more than one ion channel species, and modifications affecting sodium, potassium and calcium channels have been clearly described in recent years. Although the pharmaceutical industry has traditionally paid little attention to ion channel modulation as a potential target to control neuropathic pain, the situation is clearly changing and exciting new developments are to be expected in the near future. Voltage-gated sodium channels play a pivotal role in action potential generation and conduction and are therefore of special interest for many pharmaceutical industries.17 Indeed, several small and some big companies have sodium channel blockers in their development pipelines. Potassium channels are also of importance due to their ability to limit repetitive discharge and to set resting membrane potential, and research is also being conducted to find new molecules capable of modulating them.18
Microneurography in the Study of Neuropathic Pain Conventional electrophysiological methods are unable to record these disperse, chaotic discharges in sensory axons. Therefore, the study of positive sensory phenomena has largely relied on quantitative psychophysical tests. In animals, possible electrophysiological correlates of positive sensory phenomena have been documented in traumatic neuromas and in demyelinated nerve fibres.19,20 In experimental human volunteers, ectopic nerve impulses generated in single myelinated sensory fibres have been correlated with post-ischemic and post-tetanic paresthesias.21 However, recordings from patients suffering from neuropathic pain have been scarce, and abnormal nerve impulse activity had occasionally been recorded in polyneuropathy, amputation neuroma and Spurling and Tinel’s signs only from highly myelinated afferent fibres. Ectopic impulses from human unmyelinated C-nociceptors were simply lacking, until very recently. This constituted a clear shortcoming in the study of the mechanisms underlying neuropathic pain in humans, because ectopic discharges from C-nociceptors are probably the basis of spontaneous burning pain.
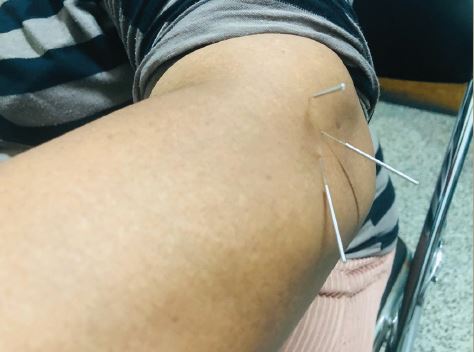
Recording spontaneous ectopic discharges in human nociceptors has been proved to be a challenging task. Identification of individual action potentials from unmyelinated C-nociceptors in humans has been possible for decades through the use of a special recording technique called microneurography. Microneurography consists of the introduction of a tungsten microelectrode within the peripheral nerve of a conscious subject.22 The procedure is minimally uncomfortable and has no long-lasting side effects. However, until recently, not much could be done in terms of a systematic analysis of the unitary or multi-unitary recordings obtained with this technique. This rather discouraging situation has begun to change due to the development of improved recording techniques and software. These improvements have allowed stable recordings from individual unmyelinated C-fibres and their segregation into different functional classes having discrete electrophysiological properties of their membranes. For example, ‘profiles’ of activity-dependent slowing in individual C-fibres of humans have been shown to correspond to specific functional types of peripheral nerve fibres.23-26 When stimulated at 2Hz for three minutes, nociceptive fibres slow by more than 10%, whereas non-nociceptive fibres slow by less than 10%. These differences indicate that some axonal membrane properties are related to the function of the nerve fibre and have allowed identification of different types of C-fibres in the human setting and the study of their behaviour in pathological states.
Targeting Membrane Hyperexcitability
Exciting new findings have been reported recently – in particular the ability to record ongoing ectopic impulse generation in C-nociceptors of patients with peripheral neuropathies and neuropathic pain (see Figure 1).9,28 This means that for the first time abnormal impulses in human nociceptors can be recorded and the effects of drugs on them can be checked. Although C-nociceptors are important, it is obvious that many diseases also affect thinly-myelinated A-delta nociceptors and large-myelinated A-beta tactile sensory units. When ectopic impulse generation takes place in these other two types of sensory unit, pain and paresthesias/dysesthesias may occur. Targeting this ectopic impulse generation with new molecules, capable of silencing or modulating it, will add new promising drugs to the armamentarium.
In summary, research into the intimate ionic mechanisms rendering membranes hyperexcitable constitutes one of the most promising fields in the development of new compounds to treat neuropathic pain. ■