Parkinson’s disease (PD) is a progressive, neurodegenerative movement disorder and the second most common neurodegenerative disorder in the United States.1 In 2020, approximately 930,000 people in the United States aged 45 years and older developed the disease, and this number is expected to rise to 1,238,000 by 2030.2 The typical age of onset is around 60 years, and the prevalence increases with age.3,4 PD results from progressive dopaminergic neuronal degeneration within the substantia nigra of the basal ganglia and the locus coeruleus. The resulting dopamine deficiency at striatum receptors interrupts signal transmission to the thalamus and motor cortex of the brain, causing the motor symptoms of PD.5
PD is perhaps best characterized by its motor symptoms. Parkinsonism is a core feature of PD and includes the triad of bradykinesia, resting tremor and rigidity, which reflects extrapyramidal system impairment. Patients often experience postural instability and shuffling gait characterized by quick and short steps and difficulty turning, making patients prone to falls.6 Patients can also display hypomimia, often described as ‘masked faces’.7
PD gravely impacts the quality of life of both patients and their caregivers. However, it is not just motor symptoms that have a profound effect on patients; non-motor symptoms, such as mood and psychotic symptoms, can also cause significant changes in patients’ emotional wellbeing and affect their and their caregiver’s quality of life.8 Nevertheless, due in part to both clinicians’ focus on motor symptoms and patients’ hesitance to report psychiatric symptoms, these symptoms are unfortunately frequently left unrecognized by clinicians, with neurologists failing to identify depression, anxiety and fatigue in at least 50% of consultations.8 This paper reviews existing literature and discusses current and emerging diagnosis and management strategies for depression and psychosis in PD. This should empower clinicians to detect these disorders early and effectively manage their patients’ treatment, so that people with PD can receive comprehensive and holistic care, and their quality of life can improve.
Depression in Parkinson’s disease
Although PD is often characterized by its motor symptoms, depression may significantly impact the quality of life of many patients with PD. Up to 35% of patients present with depression symptoms, and major depressive disorder (MDD) affects approximately 17% of patients with PD.9 While the neurochemical origins of depression in PD are unclear, the increased prevalence of depression in patients with PD may be due to the loss of dopaminergic, serotonergic and noradrenergic innervations in the limbic system and locus coeruleus, based on functional neuroimaging studies.8,10 The prevailing model explains that degeneration of mesocortical and mesolimbic dopaminergic neurons leads to orbitofrontal dysfunction, disrupting serotonergic neurons in the dorsal raphe nucleus and leading to dysfunction of the orbitofrontal–basal ganglia–thalamic circuits implicated in depression.10 In patients with PD, depression and dementia, functional neuroimaging studies have demonstrated more prominent cortical cholinergic denervation as well.10
Risk factors for depression in patients with PD include the onset of motor deficits prior to 40 years of age, increased imbalance and rigidity, cognitive impairment, chronic medical conditions, personal and/or family history of psychiatric illness and female sex.10 It is also important to consider risk factors for depression in the general geriatric population, which include bereavement, social isolation, low education level and caregiver burden.11 Depression in PD has a significant negative impact on patients’ physical and psychological quality of life, and the sequelae of depression in PD includes rapid progression of motor symptoms, accelerating cognitive decline and higher rates of disability.12 Depression in patients with PD also directly correlates with high distress scores.13 However, the duration and stage of PD does not seem to be significantly associated with depression or any domain of quality of life, whether psychological, environmental, physical or social.14
Diagnosing and screening for depression in Parkinson’s disease
A diagnosis of MDD against a background of PD requires, according to the Diagnostic and Statistical Manual of Mental Disorders, 5th edition (DSM-V), that patients have five or more of the following symptoms for at least 2 weeks, with at least one being depressed mood or anhedonia: depressed mood for most of the day, almost every day; sleep disturbance; anhedonia; feelings of worthlessness or excessive guilt; fatigue or loss of energy; decreased concentration; psychomotor changes (usually slowing, but may also be agitation); appetite changes; and/or suicidal ideation.15 As the natural progression of PD can also present these symptoms, it is sometimes challenging to distinguish whether a PD patient is depressed.16
Depressive symptoms can occur at any point during the course of PD, from the ‘pre-motor’ stage to advanced stages. A diagnosis of MDD precedes a PD diagnosis in approximately 30% of patients and a history of depression is two times more likely in the pre-motor years preceding PD diagnosis than it is after diagnosis.8,10
There are several ways to screen for depression in patients with PD. One test is the Geriatric Depression Scale (GDS-15), which is a 15 question self-rated questionnaire. It is a sensitive tool, and scores correlate well with the International Classification of Diseases, 10th Revision (ICD-10) diagnosis of depression by clinicians (p=0.00).14 In contrast to the GDS-15, the Montgomery–Åsberg Depression Rating Scale (MADRS) is rated by clinicians.17 The Hamilton Depression Rating Scale (HAMD-17) and Beck Depression Inventory (BDI) are also used, although the latter is based on the DSM, 3rd Edition (DSM-III)criteria instead of the latest DSM-V criteria.17 Due to this variety in depression scales, the Movement Disorder Society (MDS) released recommendations for screening for depression in PD. Firstly, it states that observer-rated scales are preferred and that, for screening purposes, the HAMD-17, BDI, Hospital Anxiety and Depression Scale (HADS), MADRS and GDS-15 are valid. To assess severity of depressive symptoms, the HAMD-17, MADRS, BDI and the Zung Self-rating Depression Scale (SDS) are recommended. To account for confounding factors, the Unified Parkinson’s Disease Rating Scale or other scales to assess motor symptom severity should also be used.18
Differential diagnoses of depression in Parkinson’s disease
Before diagnosing depression in PD, it is crucial to rule out other diagnoses and aetiologies that can present similarly so that appropriate treatment is offered. Familiarity with DSM-V criteria for relevant psychiatric disorders (such as anxiety and bipolar disorder) can aid in this task. Considering the older mean age of patients with PD, it is especially important to rule out mild cognitive impairment and dementia. For similar reasons, depression in PD should also be differentiated from bereavement or healthy grief reactions. Causes of secondary depression should also be elucidated through thorough laboratory and urine testing, review of systems and clinical history. Common causes/contributors include thyroid disorders, vascular disease (including stroke), malignancy, infection, chronic pain and sleep disorders.19 Medication and substance-induced depression should also be excluded. An unusual differential diagnosis that is important to carefully consider is the pseudobulbar effect. The phenomenon has a prevalence of 3.6–42.5% in patients with PD, and is characterized by episodes of unprovoked crying, or rarely laughing, that are unrelated to mood. In patients with PD, pathological crying is observed more often than pathological laughter. These symptoms may be misidentified as being mood driven, leading to a misdiagnosis of depression.20
Treatment approaches for depression in Parkinson’s disease
First-line treatment for depression in PD is usually non-pharmacologic, since many interventions are as effective as medication and do not contribute to polypharmacy in the elderly population.16 Additionally, due to the lack of large, randomized, placebo-controlled antidepressant trials in patients with PD, there is a lack of consensus regarding specific treatment response and pharmacotherapeutic treatment guidelines.16 As a result, management of depression in PD is largely a joint decision-making process involving the clinician’s evaluation of the patient and the patient’s own preferences.
Despite this lack of consensus, cognitive behavioral therapy (CBT) has the most evidence supporting its efficacy in depression in PD. CBT is the most studied therapy for depression and consists of several steps.21 First, the therapist aids the patient in identifying irrational cognitive patterns based on distorted thinking. Second, discordant emotions associated with these thoughts are identified. Third, the therapist and patient restructure thoughts, leading to positive emotional changes.21 A study by Dobkin et al. (n=80) is the largest randomized controlled trial comparing symptom severity in patients with PD with a DSM-IV diagnosis of either primary major depression, dysthymia or depression otherwise unspecified, who either received or did not receive CBT.22 The results showed that patients receiving CBT had significantly improved depression, quality of life and coping scores at 10 weeks post-treatment compared with patients who did not receive CBT. The sessions emphasized more behavioural activation and anxiety management than is typical in CBT sessions, and caregivers were also given training to encourage thought reframing when patients were out of session. All patients continued their PD medication regimen. Despite the aforementioned benefits, CBT did not have a significant effect on sleep, social support or caregiver burden, and the impact of CBT on coping and perceptions of physical disability were not significant after controlling for multiple comparisons.22 Longer-term benefits of CBT have not yet been systematically studied and may be important due to progressive neurodegeneration in PD.16
If CBT fails to ameliorate symptoms, medication should be considered (Table 1). Antidepressants have a moderate effect on symptom management, although this effect depends on medication class and is sometimes insignificant.16 Bomasang-Layno et al. conducted a meta-analysis of randomized controlled trials and found that of all antidepressants, only selective serotonin reuptake inhibitors (SSRIs) had a statistically significant effect size on depression scores in patients with PD and MDD, dysthymia or depression otherwise unspecified.23 Some studies showed that sertraline, citalopram and paroxetine may lower depression scores when compared with placebo; and venlafaxine, a serotonin–norepinephrine reuptake inhibitor (SNRI), also may have such an effect.24,25 While tricyclic antidepressants (TCAs) demonstrated a quicker response and larger effect size on depression in some studies, SSRIs are preferred, as TCAs have significant side effects, including cognitive impairment, autonomic instability, corrected QT interval (QTc) prolongation and risk of overdose.26–28 Additionally, while some studies have shown insignificant differences in depression scores with SSRIs compared with placebo, dopamine release from the anticipation of benefit can result in a robust placebo response.29 The side-effect profile of these drugs is minimal, and if pharmacologic treatment is being pursued, SSRIs should be offered first.30
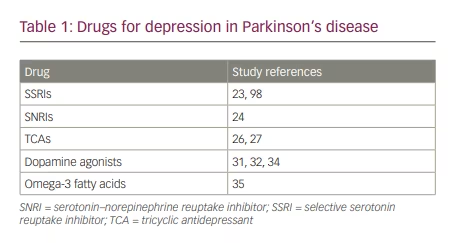
Dopamine agonists might also provide symptom relief for patients. Barone et al. conducted a randomized, double-blind placebo-controlled trial (n=296) and found that in patients with PD who had high self-rated depression severity scores, pramipexole (0.125–1.0 mg three times daily) significantly improved depression scores compared with placebo. Around 80% of the drug’s total impact on depression scores was predominantly mediated by direct antidepressant effect, while 20% was attributed to improvement in motor symptoms.31 Another study by Barone et al. was a multicentre, parallel-group, randomized non-placebo-controlled study of 67 patients with PD and MDD. They compared depression scores before and after receiving pramipexole (1.5–4.5 mg/day) or sertraline (50 mg/day) for 12 weeks. While depression scores improved in both groups, the proportion of patients who recovered from MDD was significantly higher in the group taking pramipexole than taking sertraline.32 In addition, Jiang et al. conducted a meta-analysis of 18 randomized controlled trials that showed that pramipexole (0.25–4.5 mg/day) significantly decreased SDS and/or HAMD-17 scores compared with the control group.33 More research is needed before pramipexole can be routinely recommended.31,32,34
Additionally, omega-3 fatty acids may help improve depression symptoms. Da Silva et al. conducted a randomized, double-blind placebo-controlled study (n=29) and found that patients with PD and MDD who took four capsules (each containing 180 mg of eicosapentaenoic acid and 120 mg of docosahexaenoic acid and tocopherol) of omega-3 fatty acid daily for 3 months with or without concurrent antidepressants had significantly improved scores in two out of three depression scales, compared with those who took placebo.35 Omega-3 fatty acid supplements may therefore be a benign supplement to other treatment approaches for patients with depression and PD, especially given their favourable side-effect profile and affordability.35
Apart from CBT and pharmacologic treatment options, other non-pharmacologic treatments are also being explored. One of these is aerobic exercise/resistance training, which may improve mood in patients with PD, and is often suggested as a complementary therapy to dopamine agonists and antidepressants.36–40 Wu et al. systematically reviewed randomized controlled trials or quasi-experimental designs (n=342) and found that in patients with PD, aerobic training (stretching-strengthening, walking and/or stepping for 45–60 minutes, 2–3 times a week for over 8 weeks) significantly improved depression and quality of life scores the most compared to other types of physical activity.36 While the reasons for this are unclear, murine models suggest that exercise may improve depression symptoms by upregulating brain-derived neurotrophic factor (BDNF).41 However, patients who have advanced motor symptoms may be physically limited by this approach.
Another therapy being explored is repetitive transcranial magnetic stimulation (rTMS). While more robust studies are necessary, some randomized controlled trials have shown significant improvement in depression symptoms with rTMS compared with sham rTMS.42,43 Makkos et al. conducted a randomized, double-blind placebo-controlled study (n=46) in which rTMS was applied bilaterally over the motor cortex (M1) of the brain.43 Patients with PD who received rTMS had a statistically significant improvement in depression scores and health-related quality of life, in addition to motor function. This improvement did not occur in the group treated with sham rTMS. Despite the results of this study, other randomized controlled trials have not demonstrated a significant difference between rTMS and sham TMS.44,45
Electroconvulsive therapy (ECT) may be considered if other approaches have failed, or if the situation is urgent (patient is actively suicidal) and there is no time for antidepressants to take effect.46 Although the efficacy of ECT in depressed patients without PD has been well established, no randomized controlled trials have demonstrated the benefit of ECT in depressed patients with PD.47 Still, studies suggest that ECT may ameliorate depression symptoms and also improve motor symptoms. Takamiya et al. conducted a meta-analysis (n=129) that showed that ECT significantly improved depressive and motor symptoms in patients with PD. They also demonstrated that ECT did not decrease cognitive functioning, and decreased the ‘wearing-off’ phenomenon associated with levodopa.48 Borisovskaya et al. also conducted a meta-analysis of 116 case reports, which supported Takamiya’s findings.49 Despite the conclusions from these studies, ECT is not readily given as treatment, seemingly due to stigma, patient hesitancy and the transient confusion and delirium that occur in up to 33% of patients with PD who undergo ECT.47,49
While there are no controlled trials studying its efficacy specifically in patients with PD and depression, vagus nerve stimulation (VNS) has been suggested as a possible approach for any patients with treatment-refractory depression.50 Mondal et al. conducted a randomized, double-blind, sham-controlled trial (n=33) and found that PD patients who received six two-minute stimulations of the VNS device daily had significant improvements in walking speed, stance time and step length compared to sham.51 Although this suggests that VNS is well tolerated in PD patients and may reduce motor symptoms, more research into the effect of VNS specifically in patients with PD and depression needs to be conducted before VNS can be recommended as a treatment.51
Deep brain stimulation also holds promise and may be able to be used as a last-resort treatment. It is commonly used to improve motor symptoms in patients with PD who no longer improve with medication, but studies suggest it can improve depression symptoms as well.52,53 This effect is greater when the globus pallidus internus is stimulated, and the intensity of improvement is directly proportional to both duration and severity of PD before deep brain stimulation. In these patients with advanced disease, scores evaluating both motor and non-motor symptoms in PD patients improved regardless of whether the globus pallidus internus or subthalamic nucleus was stimulated.54
As for future therapies, murine models and case reports suggest istradefylline, ketamine, BDNF, pioglitazone and melatonin are promising adjuvant treatments.55–58 More robust research is needed before they can be recommended as therapies.
Parkinson’s disease psychosis
Psychosis is important to recognize in patients with PD, and approximately 60% of patients will develop symptoms within 12 years of PD onset.59 PD psychosis (PDP) has a significant and negative impact on quality of life. Psychotic symptoms independently predict increased mortality and nursing home placement in PD.60 While dopaminergic medications for PD motor symptoms may contribute to PDP, their role in developing the disorder is hypothesized to be minimal.60,61 Instead, PDP seems to be a progression of the neurochemistry underlying PD, as PDP is associated with Lewy body pathology, monoaminergic neurotransmitter imbalances (particularly serotonin) and visuospatial processing deficits.62 The inferolateral temporal lobe has been implicated in visual hallucinations, and studies show higher densities of Lewy bodies in the amygdala and parahippocampus in patients with PD with psychosis versus without.63 Increased levels of serpin-A5 in the locus coeruleus, substantia nigra and primary motor cortex, as well as reduced trypsin-2 expression in the anterior cingulate cortex, could also be linked to PDP, but further studies to clarify this are warranted.63
Manifestations of PDP vary among patients, and symptoms tend to increase in severity as PD progresses.62 However, usually, psychosis initially presents with visual illusions and progresses to visual hallucinations with insight, visual hallucinations without insight and, lastly, delusions. Visual illusions impact 17–72% of patients with PD, and there is up to a 50% lifetime risk of developing visual hallucinations.61 Simple visual hallucinations may consist of flashes of light and blurry colours. In contrast, complex hallucinations often consist of images of people, animals or objects.64–66 They can last seconds to minutes and are more common as PD progresses.64 Most frequently, they occur when lighting is poor, such as when waking up in the morning, or in the evening.64 Table 2 summarizes these hallucination types.
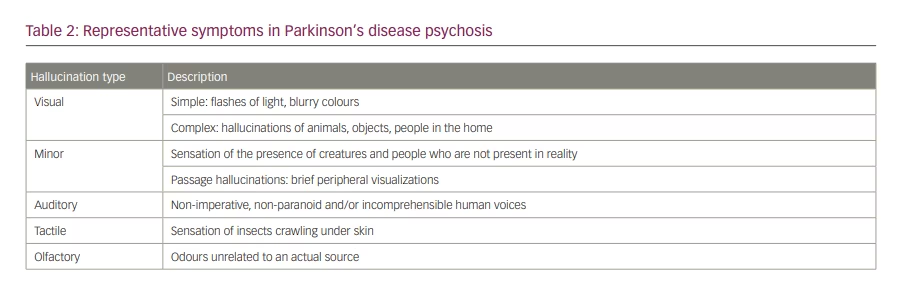
Auditory hallucinations, tactile hallucinations, and delusions are less common than visual hallucinations. If they do occur, they are associated with older age and occur together with visual hallucinations. Auditory hallucinations in this population usually consist of hearing human voices, and tactile hallucinations typically involve a sensation of insects crawling in/on the patient’s skin.67 Apart from visual hallucinations, patients can also experience minor hallucinations. One type involves sensing the presence of another person. This is most likely to occur indoors and upon awakening. Another type of minor hallucinations is passage hallucinations, which are brief peripheral hallucinations that are also more likely to occur indoors and when waking up. They are visual illusions defined as misperceptions of real external stimuli and can precede development of motor symptoms or complex visual hallucinations.68
There are some risk factors for developing PDP, though they depend on the symptom(s) the patients manifest. For example, comorbid dementia and cognitive impairment increase the odds that a patient will develop hallucinations. In fact, patients with PD and dementia are six times more likely than those without dementia to develop hallucinations.69 Additionally, patients in nursing homes are at increased risk of developing paranoia than patients not in nursing homes.69 Lastly, sleep disorders seem to be a risk factor for PDP. Pappert et al. identified 82% of patients with PDP as having a sleep disorder.70 Interestingly, daytime somnolence seems to predict the development of visual hallucinations.71
Diagnosis of Parkinson’s disease psychosis
Diagnosing PDP is not straightforward and is ultimately a clinical task. To aid clinicians, the MDS in 2008 recommended using a combination of different scales for diagnosis: the Neuropsychiatric Inventory, the Brief Psychiatric Rating Scale, the Positive and Negative Syndrome Scale, and the Schedule for Assessment of Positive Symptoms.72 Another set of criteria is the National Institutes of Neurological Disorders and Stroke-National Institute of Mental Health (NINDS-NIMH) criteria. To be diagnosed by these criteria, patients must present with at least one of the following symptoms: illusions, a false sense of presence, hallucinations or delusions that are recurrent or continuous for at least 1 month after being diagnosed with PD according to the UK Brain Bank criteria for PD.73 These criteria do not measure psychosis severity, and a rating scale must be employed to do so. The MDS has since stated that current scales are insufficient to be considered definitive rating tools and has endorsed development of a new scale to assess PDP.72
Differential diagnosis of Parkinson’s disease psychosis
To refine the diagnosis of PDP, care must be taken to exclude diagnoses that may present similarly to PDP. This can be done with a thorough history and review of systems. Some of the most important diagnoses to consider are neurodegenerative diseases. Perhaps the most important of these is Lewy body dementia (LBD), since it also can present with motor and psychotic symptoms. In contrast to PDP, psychotic symptoms in LBD are often the presenting manifestation of the disease. LBD is also more likely to present with visual hallucinations and delusions than PD with or without dementia, and psychotic symptoms instead of motor symptoms predominate.74 In PDP, the presence of hallucinations is associated with increased severity of motor symptoms.74
Other neurodegenerative diseases that should be considered are Alzheimer’s disease, vascular dementia, frontotemporal dementia (FTD) and Creutzfeldt–Jakob disease (CJD). These pathologies are more likely to present with delusions and, in FTD, negative psychotic features such as flat or blunted effect, apathy, poverty of speech and anhedonia are most common. In contrast, hallucinations are more common in PDP than in Alzheimer’s dementia, vascular dementia, FTD or CJD.75
Delirium with psychotic features is important to rule out and treat accordingly once the underlying cause is elucidated. The prevalence of delirium in patients with PD ranges from 8% to 24%, and a recent study by Lubomski et al. showed that patients with PD had a five-times higher risk of developing delirium than healthy controls.76 In contrast to psychosis in delirium, PDP is insidious in onset. Patients have clear consciousness and subtle cognitive defects. In addition, patients with PDP usually do not have acute comorbidities, and key symptoms are usually hallucinations, delusions, thought insertion (belief that one’s thoughts are someone else’s), broadcast hallucinations (belief that one’s thoughts can be heard by others), passivity phenomena (belief that one’s thoughts and/or actions are controlled by others) and phantom boarder (belief that an uninvited person is living in one’s home) as opposed to inattention, thought disorganization, and day/night reversal in delirium.77
Encephalitis, autoimmune or otherwise, can also present with psychotic symptoms, although this is rare. It is typically associated with disturbances of consciousness, speech dysfunction, seizures and/or autonomic dysfunction. These symptoms are unusual in PDP, but cerebrospinal fluid analysis can be done to rule out encephalitis.78
A familiarity with the DSM-V criteria of relevant psychiatric disorders like MDD with psychotic features and bipolar disorder with psychosis is also important, as these can look like PDP, and treatment approaches differ.16 Additionally, a urine drug screen should be performed to rule out substance-induced psychosis. These substances include amphetamines, ketamine, phencyclidine and marijuana, among others. Lastly, because medications used to ameliorate PD motor symptoms (i.e. levodopa/carbidopa, pramipexole) increase dopaminergic neuro-transmission, higher doses may cause or contribute to psychotic symptoms in some patients.79
Treatment approaches for Parkinson’s disease psychosis
After the above aetiologies are excluded, the first step should be to determine whether the patient’s symptoms are induced by dopaminergic or other medications by attempting to decrease the doses of those medications. A proposed removal sequence would start with anticholinergics, amantadine and dopamine agonists first, followed by monoamine oxidase inhibitors and catechol-O-methyl transferase inhibitors. Because PD motor symptoms often increase after these changes, adjusting the dosage of levodopa may be recommended. If psychotic symptoms persist, reducing the dosage of levodopa can be considered, but this may result in an unacceptable increase in motor symptoms.80 Non-PD medications that can cause psychosis, such as steroids, should also be eliminated from the patient’s medication regimen, if possible.68
If adjusting PD medications is unsuccessful, non-pharmacologic therapies should be considered next.68 Ensuring patients adhere to a circadian rhythm and reassuring patients that their hallucinations are not real are important measures. Improving lighting to decrease hallucination incidence and other environmental modifications may also help.81,82
If these measures do not provide adequate relief, pharmacologic approaches should be considered (Table 3). Pimavanserin, approved by the US Food and Drugs Administration (FDA) in 2016, is the first-line drug for treatment of PDP.82 It is a selective 5-HT2A inverse agonist and is administered at 34 mg a day with or without food and without titration.83 Pimavanserin has been demonstrated to be effective in many studies.84, 85 Cummings et al. conducted a randomized, double-blinded, placebo-controlled phase III trial (n=199) and showed that pimavanserin resulted in a statistically significant improvement in scores for positive psychotic symptoms, with no significant motor symptom worsening.84 Tampi et al. reviewed this and one other randomized, placebo-controlled double-blinded study and showed that 80.5% of individuals treated with pimavanserin experienced at least some improvement in symptoms compared with 58.1% of individuals who received a placebo (n=259).85 Despite demonstrated efficacy, the drug’s high cost serves as a barrier to many patients.65 Additionally, pimavanserin should be avoided in patients with severe cardiac and/or hepatic or renal impairment (creatinine clearance <30 mL/min), as it is metabolized by CYP3A4 and CYP3A5 and may result in QTc prolongation.86 It should also be used cautiously in patients with dementia, since morbidity and mortality risk is increased.83,87,88 Despite these cautions, it is worth noting though that, in 2018, the FDA stated that benefits of pimavanserin outweigh the risks, and it should be thoughtfully considered.89
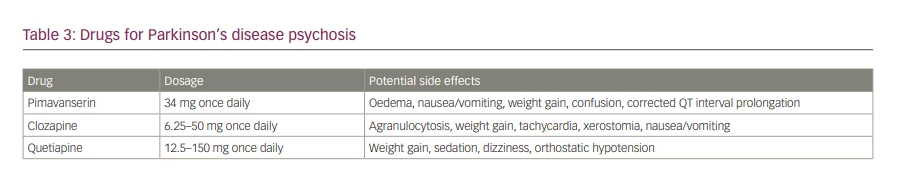
If pimavanserin fails or is contraindicated, clozapine, an atypical antipsychotic, should be considered. Recommended dosing is 6.25–50 mg per day, and it has shown efficacy at these dosages in treating PDP.90,91 While less expensive than pimavanserin, it presents with a risk of agranulocytosis, so regular monitoring absolute neutrophil counts is necessary.92 Due to this risk, many clinicians instead opt for quetiapine. Despite its use, research has shown that the efficacy in this patient population is questionable. Rabey et al. conducted a randomized, double-blind placebo-controlled study (n=58) and showed that quetiapine (mean dose 119.2 ± 56.4 mg) was not associated with a significant difference in scores for positive psychotic symptoms in patients with PDP.93 Desmarais et al. systematically reviewed randomized placebo-controlled trials (n=241) and showed that quetiapine (mean dose 103.0 mg) did not significantly improve psychotic symptom scores in patients with PDP.94 Non-placebo-controlled trials by Morgante et al. and Merims et al. have shown that quetiapine has less or the same effect as clozapine when treating PDP.95,96
Conclusion
It is well understood that the motor symptoms of PD have a profoundly negative impact on patients’ quality of life. Non-motor and behavioural symptoms can also devastate the lives of these patients and cause severe stress for caregivers. However, they may be underrecognized and untreated by clinicians.97 Depression and psychosis are common in the PD population and should be screened for at every visit, diagnosed and managed to improve the quality of life for patients with PD and their caregivers. A comprehensive differential diagnostic evaluation needs to be done to rule out alternative diagnoses and aetiologies, and familiarity with diagnostic alternatives is key. While there is a lack of consensus about how to treat these disorders, non-pharmacologic measures should be attempted first. For depression in PD, CBT is often a first-line treatment, given its demonstrated efficacy and minimal side effects.22 If CBT is inadequate, antidepressants such as the SSRIs should be initiated.16,23-29,30 ECT should also be considered as a safe and effective treatment for depression in PD.48,49
For PDP, pimavanserin is the first-line pharmacological treatment and should be used when patients have no medical contraindications or cost barriers. While more robust randomized clinical trials are necessary to further investigate other treatments, and the definition and criteria for PDP must be better elucidated, current strategies for diagnosing and managing non-motor symptoms should be used to ensure holistic care for patients with PD.









